

Various water forms with respective salinity in parts per thousands (ppt)
Yes, I know that saline water isn’t something of much concern to you as you are concerned about freshwater that’s the ONLY form of consumable water for humans and other living beings. However, extra knowledge is never harmful and you’d like to know that over 97% of water present on Earth is saline!
Out of this, the oceans have the highest level of salinity among all the major water bodies as they contain approximately 96.5% of the Earth’s water.
But have you ever wondered why it’s so?? Let’s discuss. Without going into deep, ocean water is salty because of the water runoff from the land and openings in the seafloor. But remember that various other factors too contribute to the salt content in the ocean water and other water bodies like the sea. Neither I nor you can measure how much salt is dissolved in oceans or sea, however, both of us can do so with a small water sample using a device called a salinity meter. If you want to know more about the above device, continue reading. But before that,
Let’s discern the other critical things in this regard as well like:
What is saline water?
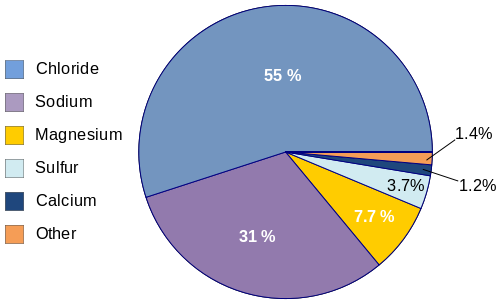
Various salt ions present in ocean water
In layman’s terms, any water that contains a significant concentration of dissolved salts like sodium chloride (NaCl) is called saline water. It’s to be noted that this concentration of dissolved salts is described in parts per million (ppm). Therefore, the following are the dissolved salts concentrations in ppm found in water forms:
● Freshwater: <1,000 ppm
● Slightly saline water: 1,000 ppm to 3,000 ppm
● Moderately saline water: 3,000 ppm to 10,000 ppm
● Highly saline water: 10,000 ppm to 35,000 ppm
● Ocean water: 35,000 ppm
Is saline water of any use?
With over 71% of the Earth’s surface enclosed with water, how come water shortages a common problem people face across the globe? Do you know why? It’s because as I said above, not all water available on Earth is usable and it’s because out of this 71% water, over 97% is saline and unfit for purposes like drinking, irrigation, and many industrial uses.
From the above data, slightly saline water is sometimes and at some places taken similarly as freshwater as its salt content is bearable, being just 1000-3000 ppm. Similarly, moderate to highly saline water has limited uses in particular industries like the thermoelectric-power industry, etc. In a nutshell, raw saline water is of no use in regular household purposes, however, if properly treated, the same saline water can be utilized in place of freshwater.
What is a salinity meter and how does it work?
Just like other handheld devices, a salinity meter can measure the concentration of dissolved salts in a wide range of water samples. They are available in the form of basic meters as well as salinity refractometer (as shown below).

Salinity refractometer
While salinity meters usually measure salt concentrations in parts per million, salinity refractometers do the same in parts per thousands (ppt). Below is a quick explanation of how a salinity meter works.
You know that salts get dissolved in water in the form of ions. Due to this, the most common way for salinity meters to measure salinity levels is through electrical conductivity, a measure of ion concentration present within the substance or water sample. Hence, measuring the salt concentration means the same as measuring the concentration of the ions. A typical salinity meter is shown below.

As you can see, there are two probes/electrodes at the bottom of the meter. As you immerse the meter in your water sample, a transfer of a current will occur between these two electrodes. As the current’s strength will depend upon the concentration of ions – the more the ions will be present, the higher the conductivity reading will be, and the higher the salinity. However, the fewer the ions present, the less conductive the substance/sample will be. Now, since we don’t need the conductivity reading, the meter’s software will automatically convert the reading into the salinity value in ppm, ppt, g/L, or mg/L using a conversion factor that’s mostly 0.5, however, not all salinity meters use the same factor.
Note: Salinity readings vary with the temperatures of the sample to be tested. Therefore, it’s suggested to use a meter with built-in automatic temperature compensation (ATC). This feature adjusts the results based on actual temperatures, thus ensuring accurate readings.
The final takeaway
Saline water, in its raw state, isn’t consumable and inappropriate for use in various industries. By the term saline water, remember that it contains dissolved ions of the salts and not the salt itself. If you wish to measure the ion concentration in any water source, you can do so using a salinity meter. Such a device uses the electrical conductivity of the water to determine the concentration of ions dissolved in it. The higher the ion concentration, the more saline it’ll be and vice-versa.
