
When you’re talking about the quality of water, you won’t mean it by its level of cleanliness your eyes can perceive. Instead, it’s the water’s pH and ORP measurements that determine the degree of quality that water has as it’s crucial to a vast number of industries like the petrochemical industry, food industry, etc. However, being a bit complicated, most of you might often confuse both the terms or assume both the two sides of the same coin. Right? To bypass this, I’ve come up with this piece to clear out all your qualms scrutinizing the pH and ORP measurements, and rest everything of your interest. So, let’s start.
What is pH?
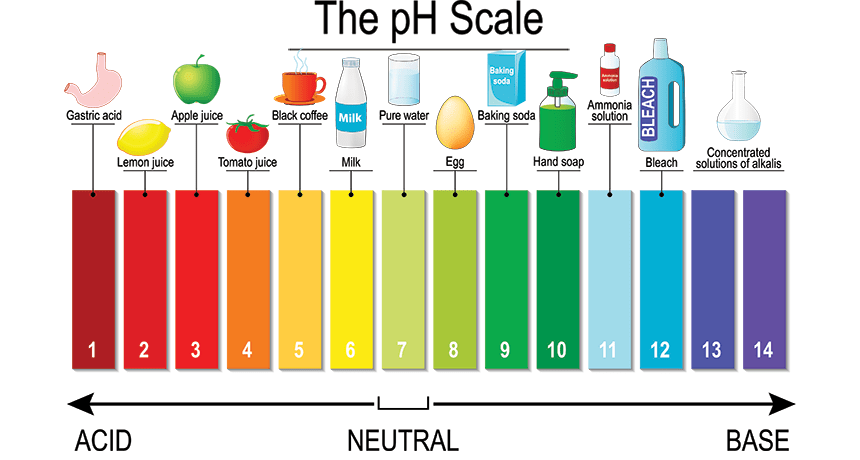
Well, a pH (power/potential of hydrogen) is a scale used to measure the acidity, neutrality, or alkalinity of a substance/solution. It’s a direct representation of the ratio of hydrogen (H+) and hydroxyl (OH-) ions in the given solution. As represented in the above figure, a pH scale is a color-gradient scale with a particular color assigned to each pH value. It ranges from 1-14 out of which:
● The more you travel towards the left of the scale, the more acidic the solution gets, and
● The more you travel towards the right of the scale, the more alkaline your testing solution gets.
That is, on a scale of 1-14, from 1-6, the solution is considered “acidic” in nature, a perfect 7 comes in the “neutral” category, and from 7-14, the solution becomes more “alkaline”.
How do you measure pH?
A pH meter is the most common device utilized for pH measurement. The working of such a device is simple to understand. As the pH of a solution is proportionate to the exchange of hydrogen (H+) ions and hydroxyl (OH-) ions, the pH meter just checks for the gain or loss of these H+ and OH- ions. Any drop or gain of these ions affects the pH of that solution and vice-versa. A pH scale is basically a logarithmic scale and its formula is:
pH = -log10[H+] or -log10[OH-]
Now, if the concentration of hydrogen ions is greater than hydroxyl ions, the pH will be <7 i.e. the solution will be acidic and vice-versa. Measuring the pH of water is critical as it helps to figure out the presence of chemical contaminants in the water and deduce its usability. For example, you can’t drink acidic water, however, the same acidic water is beneficial in facial cleansing.
What is ORP?

ORP stands for “Oxidation-Reduction Potential” and it’s a measure of the cleanliness of the water or its ability to break down the various contaminants. It’s usually measured in either volt (V) or millivolt (mV). You must know that a Reduction-Oxidation reaction or Redox reaction involves both, gain of electrons i.e. reduction, and loss of electrons i.e. oxidation. One most common example of ORP measurement is a pool. In a pool, the higher the oxidation potential, the cleaner its water. Likewise, in filthy water sources, there is a higher reduction potential than oxidation.
The following are a few examples of:
● Oxidizing liquids like aerated water.
● Strongly oxidizing liquids like chlorinated water or hydrogen peroxide solution.
● Reducing liquid like the ones having active anaerobic microbes.
How do you measure ORP?

For ORP measurement, you can simply use an ORP meter. There are two types of ORP meters: handheld meters and in-built water system meters. The above one is a simple ORP meter that goes inside the water you wish to measure the ORP of. In the above ORP meter, there are two electrodes confined inside a single unit called a combination electrode. While one electrode is called an indicator electrode and is usually made of platinum/gold/graphite, the other one is usually made from silver & silver chloride and is called a reference electrode. When the electrons present inside water contact both these electrodes, some voltage develops between them and is indicated onto the meter screen in either positive or negative volts or millivolts.
Just like the pH of water is critical to measure, its ORP levels too can’t be sidelined. To summarize, water with positive ORP values is termed healthy water, and water with negative values is unhealthy.
The Final Takeaway
So, you can now say that it’s the water’s pH and ORP values that signify the health and quality of water. However, it’s worth noting that though pH is independent of the water’s ORP values, however, ORP values vary with the water’s pH. And to measure any of these, you’d require a pH and ORP meter which you obtain at best deals at Uniglobal Business, a supplier you can trust.
